Poor product quality costs manufacturers between 5–30% of annual revenue in rework, recalls, and warranty claims, according to the American Society for Quality (ASQ).
Automated systems now cut human inspection errors by up to 85%, and AI-powered vision systems have pushed defect detection accuracy up by 30% in electronics and automotive sectors alone. Yet most operations still run inspections without a structured framework, leaving serious gaps at every production stage.
This guide explores what quality control inspection is, how it works across the manufacturing inspection process and logistics quality control, which methods apply where, and what modern systems look like in practice.
What Is Quality Control Inspection? (Core Definition and Scope)
Quality control inspection is the systematic examination of materials, components, or finished products at defined checkpoints to verify they meet specifications.
It runs across the full supply chain, from raw material intake to outbound dispatch, and feeds directly into your quality management system.
A) Inspection vs. Quality Control: Why the Distinction Matters
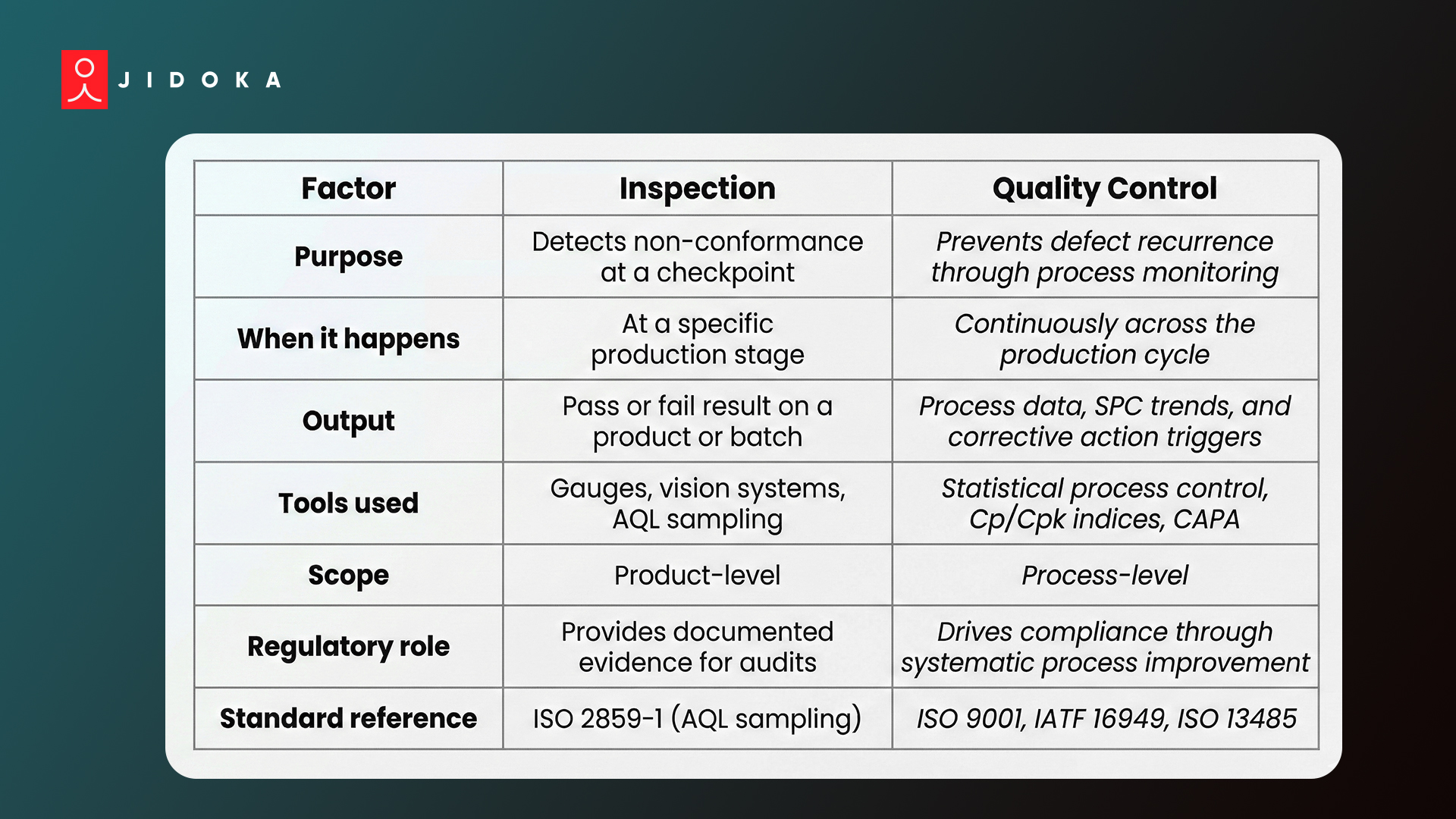
- Inspection detects non-conformance at a single point in time and records a pass or fail result
- Quality control uses statistical process control data over time to monitor process stability and prevent defect recurrence
- Without both working together, defects get caught but never stopped at the source
Inspection vs. Quality Control: Key Differences at a Glance
B) Where Quality Control Inspection Sits in a Quality Management System
- QC inspection data feeds into QA, which triggers CAPA (Corrective and Preventive Action)
- Audit-readiness depends on documented, timestamped inspection records tied to each production lot
- Regulatory frameworks like ISO 9001, IATF 16949, and FDA 21 CFR Part 820 all require structured quality control programs as a baseline compliance requirement
Understanding the definition is just the starting point. The real value shows up in how quality control inspection gets applied across each stage of the manufacturing inspection process.
Types of Quality Control Inspection in the Manufacturing Inspection Process
The manufacturing inspection process runs across three defined stages. Each stage catches a different category of risk. Skipping any one of them shifts the cost of failure further down the line, where it gets significantly more expensive to fix.
1. Incoming Inspection
Incoming inspection verifies raw materials and components against specifications before they enter production. A single non-conforming batch entering the line can trigger cascading defects across all downstream stages.
- AQL sampling under ISO 2859-1 sets the statistical basis: standard consumer product levels run at 0.0 for critical defects, 2.5 for major, and 4.0 for minor
- Supplier scorecards built from incoming inspection outcomes create an objective data trail for vendor performance reviews and source qualification decisions
2. In-Process Inspection (IPQC)
In-process inspection catches process drift before it generates large quantities of scrap or rework. It is far more cost-effective than final-stage rejection.
- Statistical process control with Cp/Cpk indices quantifies process capability in real time
- Decision logic at each checkpoint covers three outcomes: hold, rework, or scrap
- Dimensional, torque, surface finish, and weld integrity checks are the most common IPQC parameters across automotive, electronics, and industrial machinery
3. Final Inspection and Pre-Shipment Checks
Final inspection validates finished products before shipment using AQL-based random sampling at General Inspection Level II.
- Common failure categories include labeling errors, dimensional out-of-tolerance, cosmetic defect detection failures, and packaging integrity issues
- Device History Records (DHR) and batch records require inspection data that is timestamped and traceable by lot number
- Pre-shipment inspection reports should be delivered same-day to enable timely hold or release decisions before carrier booking
Each inspection stage feeds data back into your quality control program. Next covers how this extends beyond the factory floor into logistics quality control.
Quality Control Inspection in Logistics: Beyond the Factory Floor
Quality control inspection does not stop at the production line. In 2026, logistics quality control extends from supplier intake all the way through to distribution, covering every point where product condition, quantity, or traceability can be compromised.
1. Dock Receiving and Warehousing Inspection
Dock receiving inspection is the first checkpoint in logistics quality control. It verifies that what arrives matches what was ordered, in the condition it was ordered.
- Quantity verification against purchase order, packaging integrity check, and lot traceability confirmation happen at this stage
- Non-conformances at the receiving dock are a direct signal of upstream supplier issues and should trigger supplier scorecard updates
- FIFO controls must link directly to inspection status labels: quarantine, hold, or released for use
- ERP integration at receiving enables real-time quality control status visibility across procurement and production planning teams
3. Outbound and Distribution Quality Checks
Outbound checks confirm that finished, conforming products leave the facility in the right condition and with complete documentation.
- Pre-dispatch condition checks cover temperature log review, humidity verification, crush and compression testing for packaged goods, and seal integrity for food and pharma products
- Non-conformance tagging protocol requires a quarantine label applied immediately, a hold ticket raised in the QMS, and a root cause investigation initiated before the shipment clears
- Real-time ERP integration for quality control status updates prevents conforming and non-conforming inventory from mixing in transit
- Supply chain quality visibility at this stage also supports recall readiness, which regulators in the US and EU are increasingly treating as a baseline audit requirement
Strong logistics quality control programs reduce downstream warranty claims and customer complaints. Next we cover the methods and tools that make both factory and logistics quality control inspection reliable at scale.
Modern Methods and Tools Used in Quality Control Inspection
The method you pick for quality control inspection directly determines what defects you catch, how fast you catch them, and what it costs when you miss one.
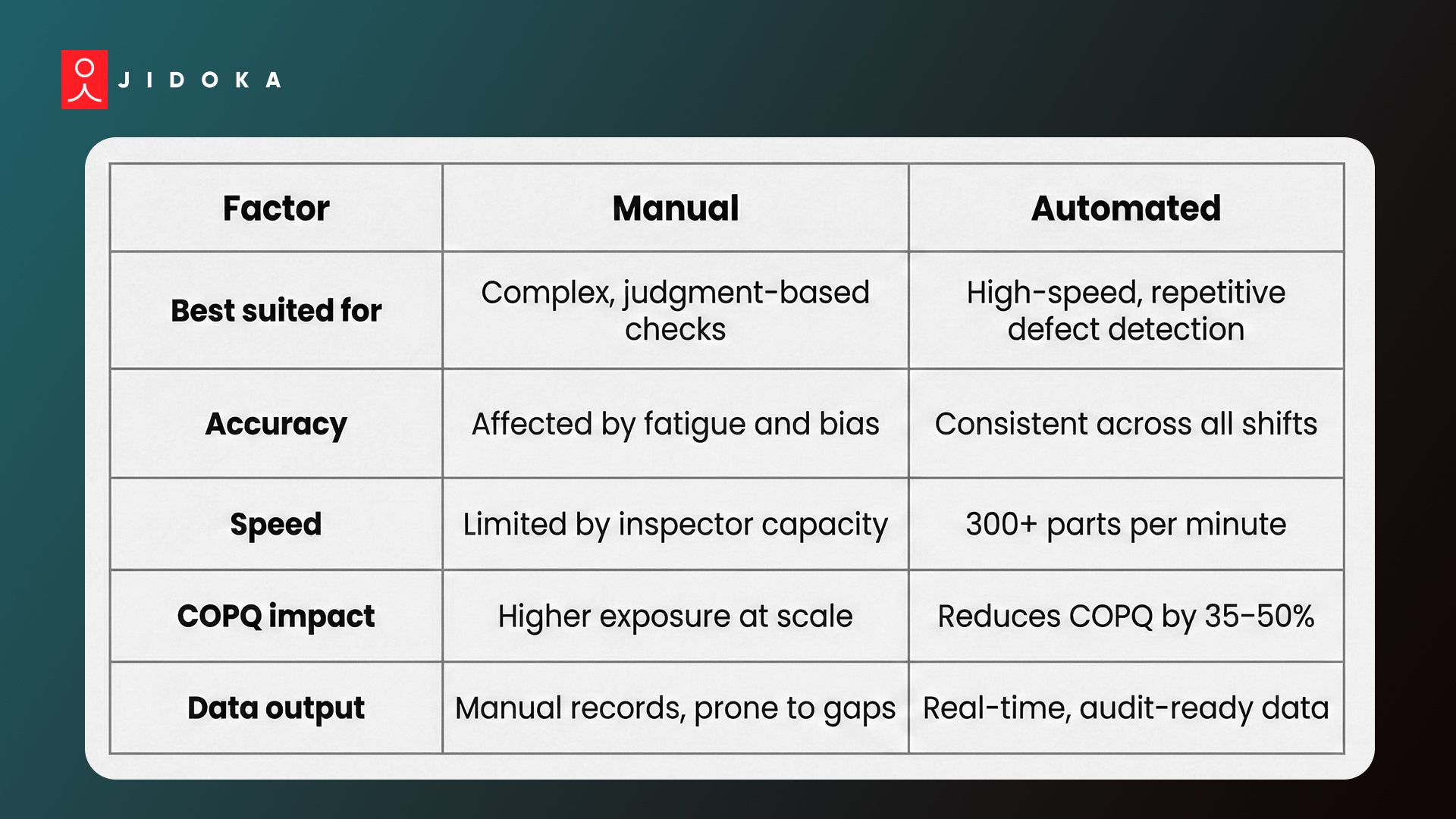
1. Manual vs. Automated Inspection: Choosing the Right Mix
- Human judgment works best for assembly sequencing verification, packaging aesthetics, and complex multi-attribute checks
- Computer vision and machine learning deliver consistent results in high-speed, high-volume production lines
- According to ASQ, average manufacturers carry cost-of-poor-quality (COPQ) at 15–20% of total sales revenue. World-class operations hold it below 5%. That gap is exactly what the right inspection mix closes
- Preventive quality control measures reduce COPQ by 35–50% when properly implemented
Manual vs. Automated Inspection: Quick Comparison
2. Non-Destructive Testing (NDT) Methods
The non-destructive testing market was valued at $22.86 billion in 2025, with manufacturing holding the largest vertical share at 29.9%.
- Ultrasonic testing leads with 25.6% market share, followed by radiographic, magnetic particle, and dye penetrant testing
- Aerospace uses NDT for weld integrity and composite structures; pharma relies on X-ray seal verification; food processing uses it for foreign body detection
- NDT replaces AQL sampling entirely for safety-critical parts in aerospace and medical devices, where a single undetected defect creates regulatory and liability exposure
Picking the right tools is half the work. The other half is knowing where your quality control inspection program is most likely to break down.
Common Quality Control Inspection Failures and How to Fix Them
Most quality control inspection programs fail for one reason: they are built as end-of-line checks rather than embedded process controls. That structural gap multiplies defect detection failures and drives COPQ higher at every stage.
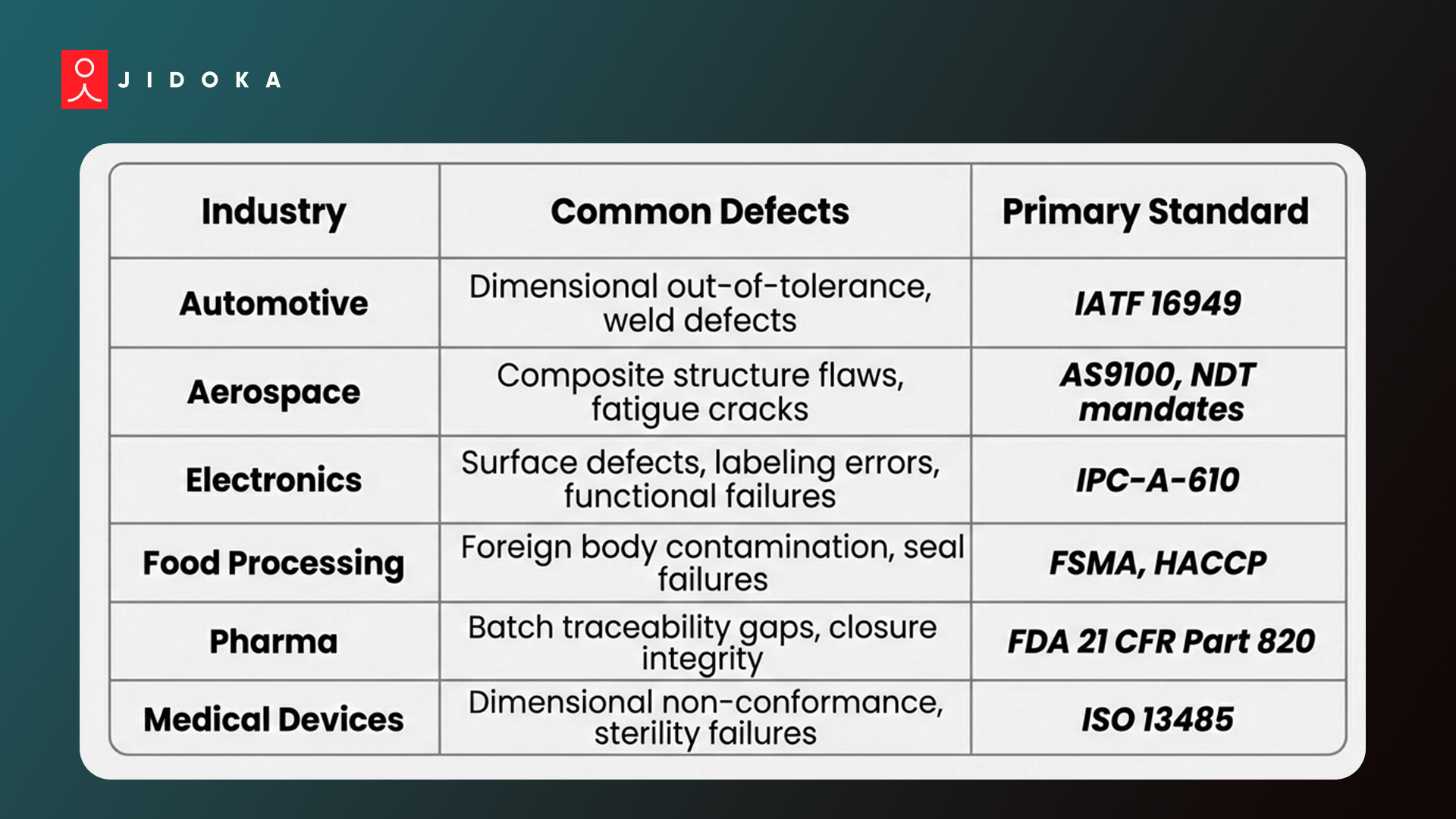
1. Top Defect Categories by Industry
- Automotive and aerospace: dimensional out-of-tolerance on structural components and weld defects flagged through non-destructive testing
- Electronics and consumer goods: cosmetic surface defects, labeling errors, and functional test failures
- Food and pharma: contamination, seal failures, and batch traceability breakdowns
- Medical devices: dimensional non-conformance on implantable components, each carrying ISO 13485 or FDA 21 CFR Part 820 CAPA obligations
Top Defect Categories by Industry: Quick Reference
2. Root Cause Analysis Tied to Inspection Data
Inspection data only creates value when it feeds back into the process.
- Fishbone diagrams categorize causes; 8D reports drive supplier-facing corrective action
- SPC trend charts monitor ongoing process stability between inspection cycles
- ISO 13485 and IATF 16949 both require documented, closed-loop CAPA programs where inspection records serve as the evidentiary foundation
- AI/ML systems analyzing historical inspection data can now forecast deviations before they breach control limits
Knowing where failures happen is only useful if your inspection system is built to act on that data fast.
How Jidoka Technologies Runs Quality Control Inspection at 300 Million Checks a Day
Most quality control inspection systems struggle under real production pressure. Jidoka Technologies builds AI-powered machine vision inspection systems that do not.
Our team aligns cameras, lighting, PLC timing, and edge units so the system performs consistently across all shifts, even at 12,000+ parts per minute.
Our Special Capabilities:
1. KOMPASS: High-Accuracy Inspector
- Reaches 99.8%+ accuracy on live manufacturing inspection lines
- Reviews each frame in under 10 ms for real-time defect detection
- Learns new variants with 60–70% fewer samples
- Handles reflective metals, printed surfaces, and textured parts
2. NAGARE: Process and Assembly Analyst
- Tracks 100% of assembly steps through existing cameras
- Flags missing parts or wrong sequences in real time using visual inspection automation
- Cuts rework by 20–35%, directly reducing cost-of-poor-quality (COPQ)
If your current quality control inspection setup cannot keep pace with your line speed or audit requirements, connect with Jidoka Technologies for a deployment assessment.
Conclusion
AI is changing how quality control inspection works across the manufacturing inspection process. Systems now detect defects faster, flag process drift earlier, and generate audit-ready data without manual intervention.
But most plants still run fragmented inspection programs, with gaps between incoming, in-process, and final stages that go unaddressed.
Those gaps compound fast. Missed defects reach customers. Recalls follow. Regulatory audits expose undocumented processes.
Operations that delay structured quality control inspection will carry the cost. Jidoka Technologies helps you identify exactly where your current inspection workflow breaks down and where automation closes it.
Let's connect with Jidoka and find out where your quality control inspection program has gaps before your next audit does.
FAQs
1. What is quality control inspection in manufacturing?
Quality control inspection in manufacturing is the systematic examination of raw materials, components, or finished products at defined stages of the manufacturing inspection process to confirm they meet specifications before moving forward or shipping to customers.
2. What is AQL in quality control inspection?
AQL (Acceptable Quality Limit) defines the maximum defect rate considered acceptable in a sampled batch during quality control inspection. Based on ISO 2859-1, it sets the statistical basis for how many units to inspect and how many failures trigger a lot of rejection.
3. What is the difference between quality control and quality assurance?
Quality control inspection identifies non-conforming products during or after production using defect detection tools and AQL sampling. Quality assurance focuses on process design and prevention, ensuring the conditions that produce defects are removed before they occur within the manufacturing inspection process.
4. How does AI improve quality control inspection?
AI-powered machine vision inspection systems analyze products in real time, flagging dimensional errors, surface defects, and labeling issues faster than manual inspection. Automated defect detection improves accuracy by up to 30% in electronics and automotive manufacturing inspection process lines, directly reducing cost-of-poor-quality (COPQ).
5. What standards govern quality control inspection in manufacturing?
Key standards governing quality control inspection include ISO 9001 for general quality management, IATF 16949 for automotive, ISO 13485 for medical devices, and FDA 21 CFR Part 820 for regulated pharmaceutical and medical manufacturing inspection process environments.








